ABSTRACT
Bilateral diaphragm paralysis due to bilateral isolated phrenic neuropathy (BIPN) is a very rare cause of unexplained respiratory failure.
We present a 65-year-old patient with no relevant previous medical history who presented in the Pulmonology Clinic with mMRC1 dyspnoea and orthopnoea. After the medical work-up, diaphragmatic paresis was diagnosed. Inspiratory muscle training resulted in mild symptomatic improvement and treatment with noninvasive mechanical ventilation (NIV) was initiated.
This condition is generally chronic and has a poorer prognosis, compared to other cases of phrenic nerve involvement. In this case, NIV restored near-normal daily function.
LEARNING POINTS
- Besides being a rare cause of dyspnoea, diaphragmatic dysfunction should be considered in the medical evaluation.
- Noninvasive mechanical ventilation is the mainstay of treatment for symptomatic patients.
- Neurologic and electrodiagnostic evaluation is essential, since the differential diagnosis of phrenic nerve dysfunction may imply different treatment strategies.
KEYWORDS
Diaphragmatic paralysis, dyspnoea, hypoventilation, noninvasive ventilation, phrenic neuropathy
INTRODUCTION
Diaphragmatic dysfunction is an uncommon and often misdiagnosed cause of dyspnoea[1].
The diaphragm is the major muscle responsible for breathing. It is a thin, dome-shaped striated muscle that separates the abdominal and thoracic cavities. It is primarily innervated by the phrenic nerve which is formed from branches of the cervical nerves C3, C4 and C5. Diaphragmatic dysfunction can be caused by diseases that affect the central and peripheral nervous system, neuromuscular junction and muscle[1].
In patients with low lung volumes on imaging examinations, hypercapnia and orthopnoea diaphragmatic paralysis should be considered. The pulmonary function tests (PFT) reveal a restrictive pattern and maximal inspiratory pressure (MIP) is reduced[2].
In symptomatic patients with bilateral diaphragmatic paralysis, noninvasive mechanical ventilation (NIV) can be an effective therapy regardless of spirometric or blood gas deterioration[3]. Other therapeutic options for diaphragmatic dysfunction are respiratory muscle training[4] and plication of the diaphragm (only in unilateral disease)[5].
CASE DESCRIPTION
A 65-year-old ex-smoker, male, with no relevant previous medical history, including for thoracic trauma, referred to the Pulmonology Clinic due to a history of orthopnoea and mMRC1 dyspnoea, with an acute onset 1 year previously. Besides a history of nocturnal episodes of numbness of the right hand, he denied persistent numbness or weakness of the upper limbs or the presence of shoulder or cervical pain at the onset of his respiratory complaints. He underwent ambulatory thoracic radiography that showed low lung volumes and bilateral diaphragmatic elevation (Fig. 1). At the physical examination, he had desaturation while supine (SpO2 93%) and normal saturation (SpO2 99%) while standing. The pulmonary auscultation revealed bibasal muffled sounds with no adventitious sounds.
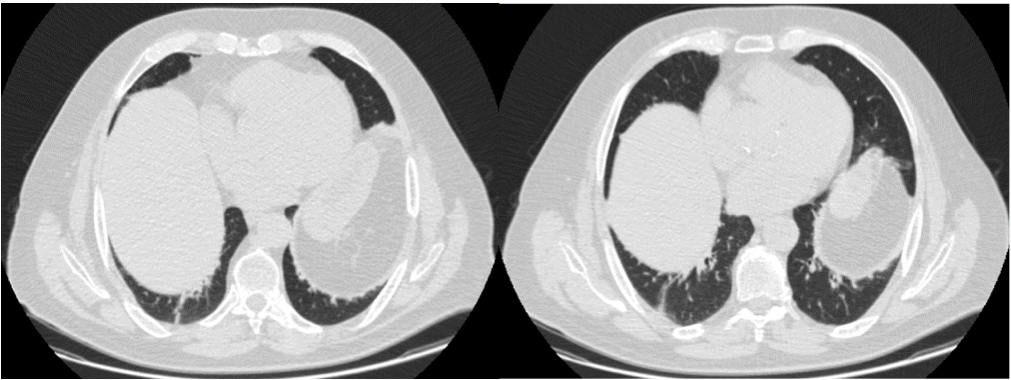
The thoracic CT scan showed symmetric elevation of both hemidiaphragms (Fig. 2), leading to decreased expansibility in the basal lungs and consequent atelectasis, and vascular and broncohilar crowding. There were no lung lesions.
The ambient arterial blood gas was normal. Spirometry showed a restrictive pattern (Fig. 3): functional vital capacity (FVC) of 1.92 litres corresponding to 51.9% of the value predicted for race, height, sex and age; forced expiratory volume (FEV1) of 1.5 litres (51.9%); FEV1:FVC ratio of 0.79; total lung capacity (TLC) of 78% predicted; residual volume (RV) of 124% predicted; and MIPs of 43% predicted.
Figure 1. Posteroanterior and lateral chest radiography, revealing low lung volumes and symmetrical elevation of both hemidiaphragms
Figure 2. Chest CT scan, showing elevation of both hemidiaphragms. This examination did not show any findings suggestive of other diseases
Figure 3. Flow volume curve for the patient, showing a restrictive pattern
Blood analysis showed a normal erythrocyte sedimentation rate and normal values for rheumatoid factor, antinuclear antibody and angiotensin-converting enzymes. Screening for HIV and Treponema pallidum infection was negative.
The neurography of the phrenic nerve showed absent responses on the left and severely reduced compound muscle action potentials (CMAP) on the right with moderately increased latencies. Needle electromyography of the diaphragm was not performed.
Electroneuromyography revealed signs compatible with mild carpal tunnel syndrome and excluded the presence of other mononeuropathies in the upper limbs, brachial plexopathy, polyneuropathy, motor neuropathy and myopathy. Repetitive nerve stimulation excluded significant end plate dysfunction.
DISCUSSION
Diaphragmatic paralysis may be unilateral or bilateral. When paralysis is unilateral, the contralateral hemidiaphragm assumes the work of breathing.
The most frequent aetiologies include: viral infection (Varicella Zoster, HIV, poliomyelitis), external or surgical trauma (cardiac surgery), paraneoplastic syndrome, mediastinal and breast tumours, radiotherapy, large vessel vasculitis, inflammatory myopathies, dermatomyositis or polymyositis and neuromuscular disease[6]. In the latter group, diaphragmatic dysfunction may be the first presentation of the disease. In Parsonage-Turner syndrome, bilateral involvement of the phrenic nerve occurs in 1% of patients, but is usually associated with the classic onset of shoulder pain and clinical or neurophysiological signs of brachial plexitis or multiple neuropathies involving the upper limbs. We diagnosed our patient as having bilateral isolated phrenic neuropathy (BIPN).
Since extensive evaluation excluded systemic disease and he did not manifest pain at the onset, he did not develop significant recovery, as is usual in Parsonage-Turner cases of phrenic nerve involvement, and electroneuromyography did not find involvement of other nerves[7].
Our patient presented with dyspnoea that was more severe in decubitus, which is a typical symptom of the disease. His examinations showed typical findings, which were low lung volumes with an elevated diaphragm in imaging assessments, a restrictive pattern in spirometry tests and a reduction in the MIPs. In some cases, hypercapnia can be observed by blood gas measurement[2].
In our case, electromyography of the diaphragm was not performed due to the risk of pneumothorax and lack of experience with this technique in our laboratory. In addition, the severely low-amplitude phrenic nerve CMAP obtained would hardly be related to a myogenic disorder limited to the phrenic nerve (our experience is that even in the presence of a severely affected diaphragm in the context of a systemic myopathy, the amplitudes of phrenic nerve CMAP are never strongly reduced).
The main goal of the treatment is the maintenance of adequate ventilation in order to avoid the consequences of chronic hypoventilation.
Unilateral diaphragmatic paralysis may be asymptomatic with no need for treatment. However, in the case of BIPN, NIV is often needed (mainly during sleep), as a progressive and irreversible respiratory failure may occur after fatigue of the accessory muscles[8].
NIV during the night may lead to the improvement of ventilatory function and to the correction of blood gas abnormalities. When PaCO2>45 mmHg or SpO2 during overnight oximetry <88% there is formal indication to start NIV[9].
BIPN cases reported so far seem to have poor recovery of diaphragmatic function as opposed to other causes of inflammatory or viral phrenic neuropathy, where spontaneous recovery associated with terminal and collateral reinnervation of the diaphragm is common[10,7].
Our patient underwent respiratory muscle training with some improvement of the symptoms. After that we prescribed NIV with BiPAP® in the nocturnal period, which is a comfortable and practical ventilator modality.
CONCLUSIONS
The use of MS-based proteomic analysis allowed the precise diagnosis and typing of AApoAIV amyloidosis in this patient. This is a rare type of systemic amyloidosis. It involves the heart, with indistinct clinical and histopathological features, and the kidneys with distinct presentation characterised by slowly progressive renal dysfunction, no significant proteinuria and amyloid deposition restricted to the renal medulla. A high degree of suspicion is critical to diagnose rare diseases with unknown prevalence and risk factors. In this case, cardiac biopsy and MS-based proteomic analysis were crucial for the accurate diagnosis of the underlying amyloidogenic protein and for further treatment strategy establishment. Given the possibility of codeposition of different amyloid precursors, tissue demonstration of amyloid deposition is necessary for diagnostic confirmation of the disease.