ABSTRACT
Introduction: Myasthenia gravis is a rare autoimmune disease caused by autoantibodies directed against the synapses of the neuromuscular junction.
Patient and methods: We report the case of a young patient with myasthenia gravis associated with Hodgkin's lymphoma.
Results: A 22-year-old patient presented with a 2-month history of severe weakness associated with muscle fatigability and intermittent ptosis without dysphonia or respiratory signs. Clinical examination revealed generalized myasthenia. The EMG did not show post-synaptic block, and anti-acetylcholine receptor antibodies were elevated at 3 nmol/l (normal ≤0.3 nmol/l). CT of the thorax showed an anterior medial mass. Immunohistochemistry of the mass revealed mixed cellularity Hodgkin’s lymphoma.
Conclusion: The association of lymphomas with myasthenia gravis has been rarely reported.
LEARNING POINTS
- Myasthenia gravis is an autoimmune disease of the neuromuscular junction and can be associated with malignant haemopathy.
- The coexistence of myasthenia and Hodgkin's lymphoma is very rare.
- This association is a therapeutic challenge.
KEYWORDS
Myasthenia gravis, paraneoplastic, Hodgkin's lymphoma
INTRODUCTION
The association of myasthenia gravis with another tumour, in particular intrathymic or extrathymic lymphoproliferation, is rarely reported. The causal link between these two pathologies has not yet been elucidated. We describe a patient who presented with myasthenia gravis associated with Hodgkin's lymphoma. To our knowledge, only five such cases have been reported in the literature to date.
CASE DESCRIPTION
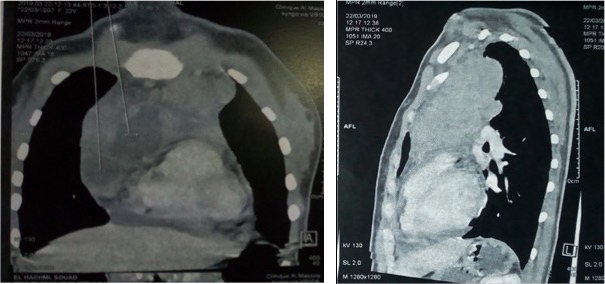
A 22-year-old patient, with no relevant medical history, presented with a 2-month history of profound asthenia associated with muscle fatigue and intermittent ptosis, without dysphonia or respiratory signs. She did not have fever, weight loss or night sweats. Clinical examination revealed generalized myasthenia gravis with a myasthenic facies associated with motor deficit. EMG did not show post-synaptic block. Anti-acetylcholine receptor antibodies were elevated at 3 nmol/l (normal ≤0.3 nmol/l). Inflammatory markers were also elevated: CRP was 45 mg/l and ESR was 35 mm/hr. The haemoglobin level was 11.6 with lymphopenia of 880/mm3. The chest x-ray revealed a medial mass. CT of the thorax revealed an anterior mediastinal mass, suggesting a thymoma (Fig. 1), the biopsy of which suggested an inflammatory pseudotumor. Two weeks later, the patient presented with a left cervical mass associated with compressive lymphadenopathy.
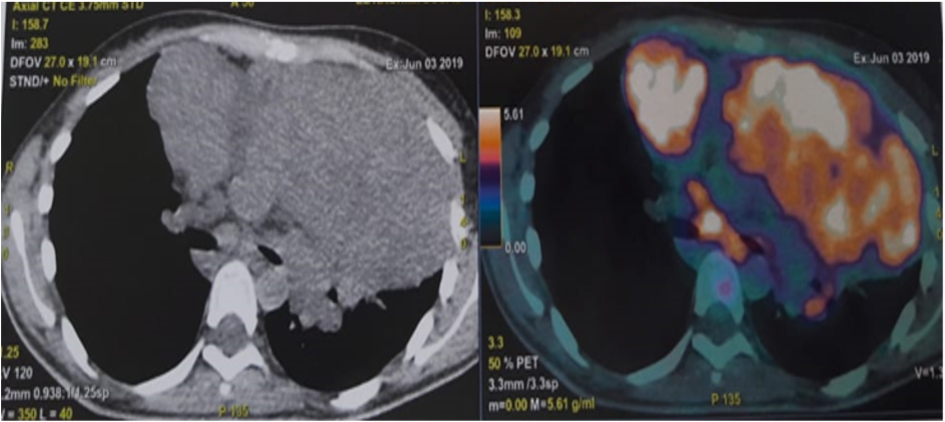
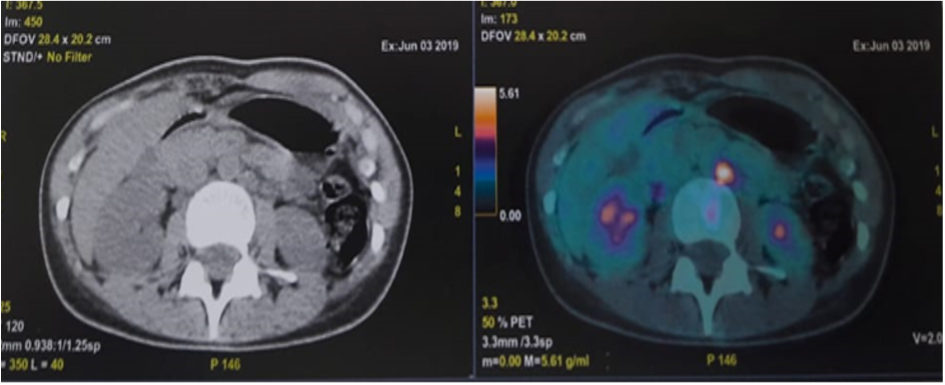
A second biopsy was performed and pathology and immunohistochemistry indicated mixed cellularity Hodgkin's lymphoma. Thus, the diagnosis of an inaugural paraneoplastic myasthenia gravis of Hodgkin's disease was made. A PET scan revealed a hypermetabolic anterior mediastinal mass associated with supra and subdiaphragmatic lymphadenopathy (Figs. 2 and 3).
Figure 1. CT of the thorax showing an anterior mediastinal mass encompassing large vessels and pushing back the left lung
Figure 2. Latero-aortic lymphadenopathy
Figure 3. Anterior mediastinal mass with lymphadenopathy
DISCUSSION
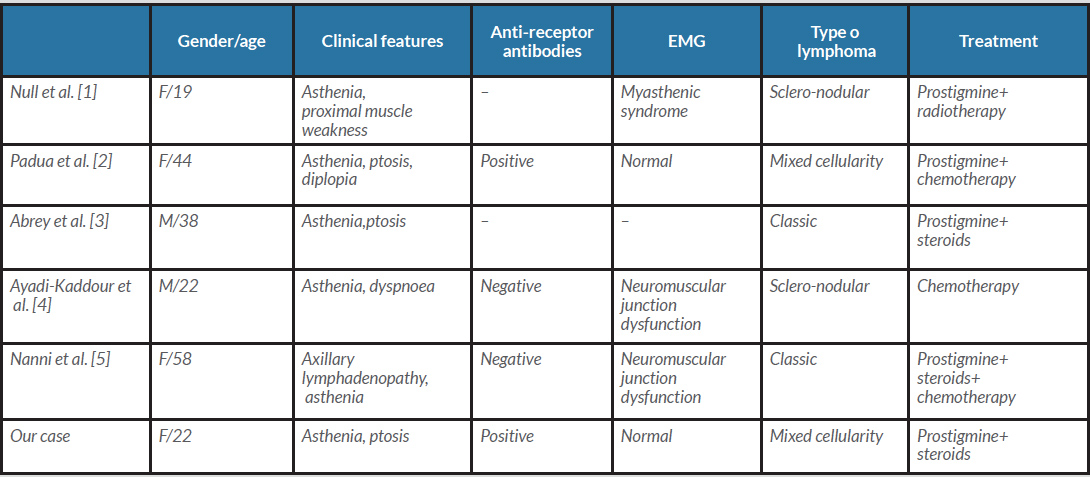
Myasthenia gravis is characterized by involvement of the neuromuscular junction. It is manifested by a myasthenic syndrome consisting of asthenia which can be complicated by dysphonia and respiratory disorders that may be life-threatening. Almost 85% of patients express anti-acetylcholine receptor antibodies. Myasthenia gravis is strongly linked to thymomas, and 10% of patients with a thymoma have associated myasthenia gravis. The association of lymphomas with myasthenia gravis has been rarely reported, and mainly concerns non-Hodgkin's malignant lymphoma. To our knowledge, only five cases of myasthenia gravis associated with Hodgkin’s lymphoma have been reported in the literature [1–5] (Table 1).
In 1977, Null et al. described the first observation associating myasthenia gravis and Hodgkin's lymphoma in a 19-year-old patient who reported asthenia with muscle fatigue [1]. The clinical examination did not reveal peripheral lymphadenopathy, while EMG confirmed involvement of the neuromuscular junction. Biopsy of an anterior thymic medial mass suggested sclero-nodular Hodgkin’s lymphoma. In 2018, Nanni et al. reported the fourth observation of this association in a 58-year-old woman with axillary lymphadenopathy, with biopsy confirming the diagnosis of Hodgkin's lymphoma [5]. A month later the patient reported asthenia with muscle weakness. Antibodies to acetylcholine receptors were negative. The EMG indicated synaptic damage. The outcome was good on chemotherapy combined with prostigmin.
A genetic hypothesis has been put forward to explain the simultaneous development of an immunological disorder and lymphoproliferation. The severity of muscle signs depends on the stage and progression of the lymphoma, so for most cases of paraneoplastic myasthenia gravis associated with lymphoproliferation, clinical remission is usually achieved after initiation of cancer chemotherapy, with a noticeable reduction in anti-acetylcholine receptor levels after treatment. However, in some patients, the severity of muscle damage, particularly respiratory and cardiac, affects therapeutic management and may be responsible for a delay in the initiation of chemotherapy.
Note that in our patient the diagnosis of Eaton-Lambert syndrome was rejected due to the patient’s age, progressive clinical signs, and absence of pulmonary involvement.
CONCLUSION
Myasthenia gravis is a very rare complication of Hodgkin’s lymphomas. Prognosis depends on rate of progress, haemopathy stage, and severity of neuromuscular damage.