ABSTRACT
Paecilomyces species are a rare cause of fungal pulmonary infections in immunocompromised hosts. Paecilomyces variotii and Paecilomyces lilacinus are the two most common species of clinical significance. Both species differ in their susceptibility to antifungal agents. We present a rare case of a 47-year-old immunocompromised woman who developed Paecilomyces pneumonia despite being on voriconazole prophylaxis. We also present a review of the literature on other reported cases.
LEARNING POINTS
- Paecilomyces species differ in their susceptibility to antifungal agents.
- Breakthrough invasive mould infections are increasingly being documented in immunocompromised patients on voriconazole prophylaxis.
- Posaconazole has been shown to be effective against both Paecilomyces species.
KEYWORDS
Paecilomyces, pneumonia, voriconazole, resistance, posaconazole
INTRODUCTION
Paecilomyces, a saprophytic fungus that lives in decaying plants, soil, wood and food products, is of great interest to clinicians worldwide due to its pathogenicity and resistance to some antifungal agents [1]. Voriconazole has demonstrated in vitro activity against many clinically significant fungi in the past, thereby validating its use for prophylaxis in immunocompromised individuals [2]. However, there has been an increase in the number of cases of Paecilomyces infection failing voriconazole treatment or prophylaxis. In this case report, we describe an immunocompromised patient with history of cutaneous T-cell lymphoma status after stem cell transplantation (complicated by graft versus host disease) who developed Paecilomyces pneumonia while on voriconazole prophylaxis. Our review of the literature demonstrates four other cases of Paecilomyces infection that either failed or were resistant to voriconazole.
CASE DESCRIPTION
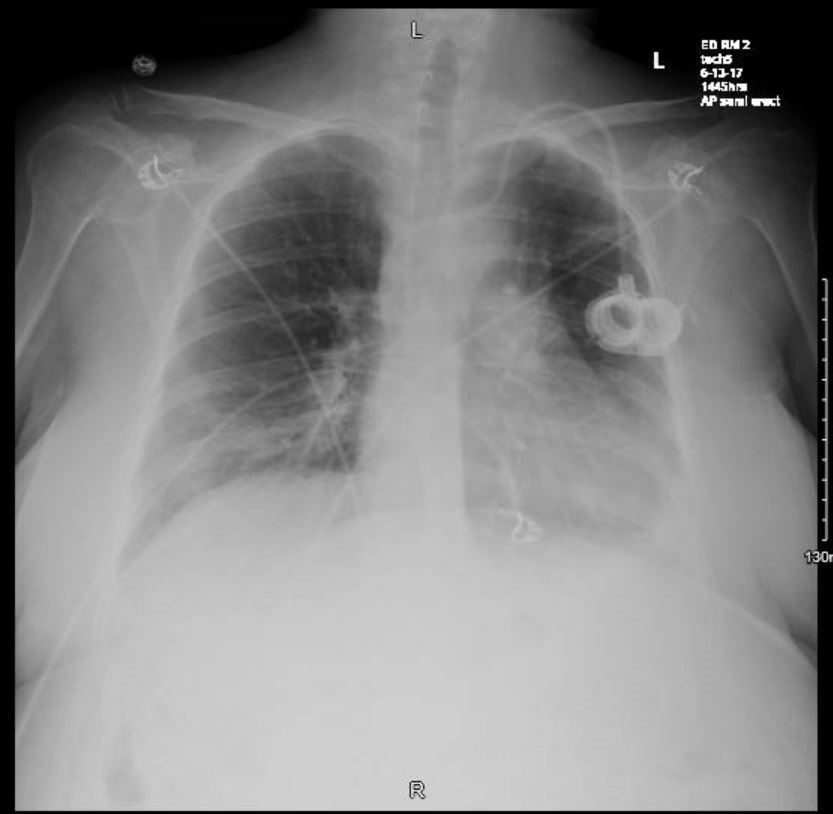
A 47-year-old woman presented to the emergency department with complaints of fever, shortness of breath, non-productive cough and generalized weakness for the last couple days. Her past medical history was significant for cutaneous T-cell lymphoma status after stem cell transplantation, graft versus host disease on photopheresis, chronic pancytopenia, secondary adrenal insufficiency, steroid-induced hyperglycaemia, hypertension and depression. Her anti-infective medications included prophylactic azithromycin, voriconazole and valacyclovir. She initially presented with a complaint of shortness of breath. Chest x-ray revealed opacities in the left lower lobe and lingula, peri-bronchial nodular opacities and a small left pleural effusion. She was diagnosed with hospital-acquired pneumonia and treated with vancomycin and piperacillin-tazobactam. She had also tested positive for rhinovirus and enterovirus. She was discharged but returned back to the hospital with tachycardia and persistent tachypnoea. Peripheral oxygen saturation on room air was 91%. Physical examination revealed bilateral crackles in the lower lung lobes on auscultation along with 2+ bilateral lower extremity oedema. Laboratory evaluation showed pancytopenia (leukocyte count of 2.67×103/µl, absolute neutrophil count of 1.73×103/µl, red blood cell count of 2.71×106/µl, platelet count of 27×103/µl), elevated C-reactive protein (CRP; 13.66 mg/dl), elevated procalcitonin (0.59), hypoalbuminaemia (2.5 g/dl) and serum (1→3)-β-D-glucan levels of 47 pg/ml. Urinalysis was remarkable for 1+ protein. Blood cultures from the prior admission grew yeast, initially identified as non-Candida species. A chest x-ray (Fig. 1) showed ill-defined airspace opacity projecting over the right lower lung, unchanged ill-defined lingular and left lower lobe opacities, and a persistent small left pleural effusion.
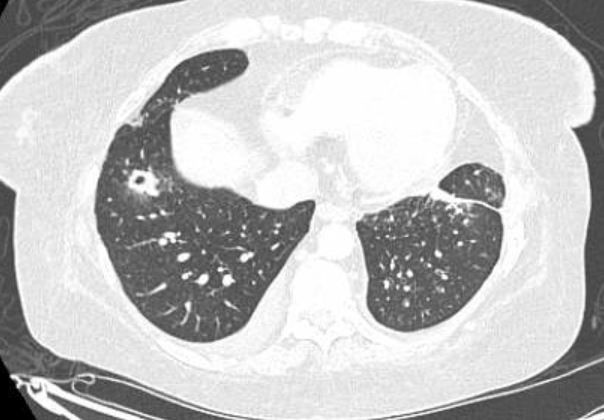
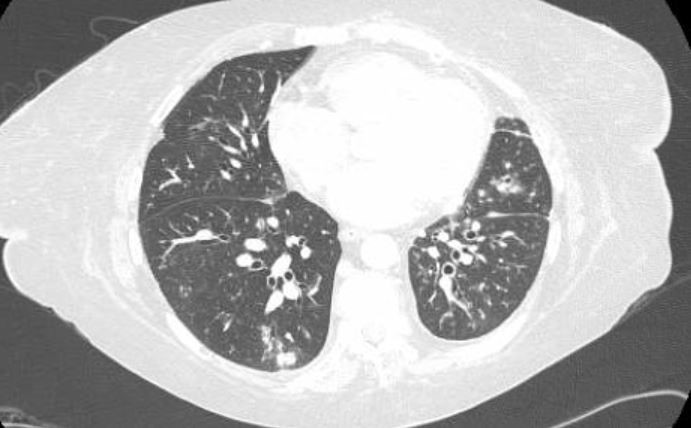
A diagnosis of hospital-acquired pneumonia was made and antibiotic therapy with vancomycin and meropenem was started. The patient’s home prophylactic medications (azithromycin and valacyclovir) were continued. The patient’s mediport was removed and she was started on IV micafungin for fungal coverage. A chest CT scan (Fig. 2) revealed patchy ground glass opacities in the right upper lobe (RUL) and right lower lobe (RLL), centrilobular micronodules and tree-in-bud opacities in the RUL, calcified granuloma in the left upper lobe, a parenchymal scar in the left lower lobe and a tiny right pleural effusion.
Figure 1. Chest x-ray showing right lower lung and left lower lobe opacities along with a small left pleural effusion
Figure 2 C.T scan of the chest showing right upper lobe patchy ground glass opacities and centrilobular micronodules, and a tiny right pleural effusion
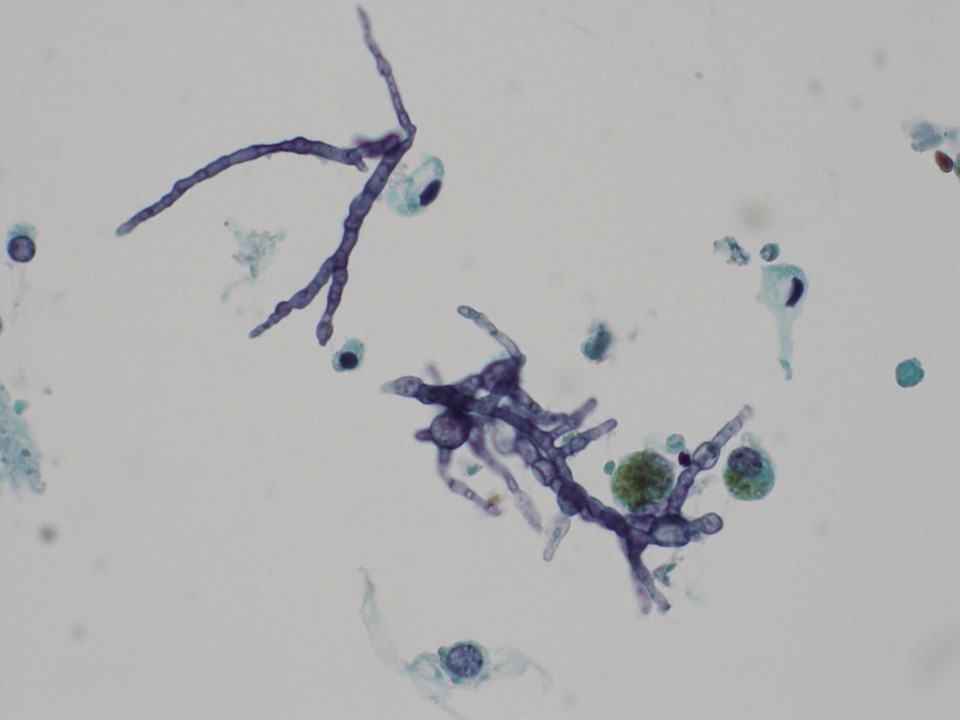
A bronchoscopy was performed and the bronchoalveolar lavage fluid culture isolated Paecilomyces (Fig. 3). A repeat CT of the chest (Fig. 4) a week later showed a small 19 mm broncho-centric cavitary nodule in the RLL (unchanged) with a new 9 mm cavitary nodule in the RLL, improved but persistent ground glass opacities and unchanged small pleural effusions. The patient was then switched from micafungin to IV amphotericin B.
During her hospital stay, the patient improved clinically with resolution of her symptoms. CRP continued to decrease with a repeat value of 0.95 mg/dl. A follow-up CT of the chest 2 weeks later showed a slight decrease in the size of the cavitary lesion but overall worsened nodularity and ground glass opacities within multiple lobes. Pleural effusions had resolved. The patient was discharged on a planned 6-week course of once-daily IV liposomal amphotericin B.
Figure 3. Irregularly branched conidiophores, swollen phialides, and ellipsoid conidia of Paecilomyces (400×)
Figure 4. CT of the chest showing cavitary nodules in the right lower lobe
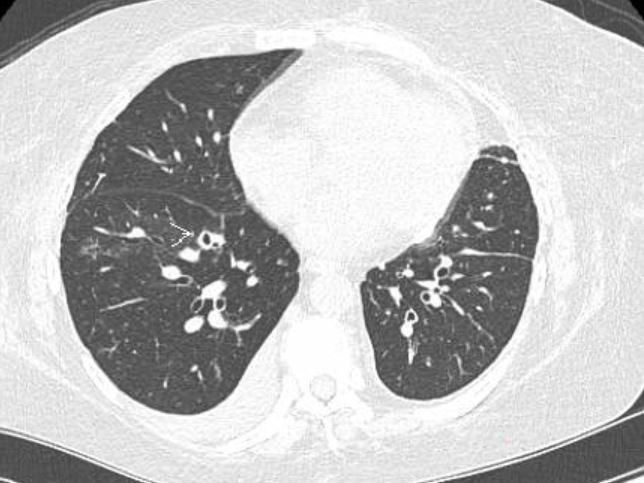
Five days after discharge, the patient was readmitted with headache and generalized weakness. Neurological evaluation including a lumbar puncture was unremarkable. Laboratory evaluation showed increasing serum (1→3)-β-D-glucan levels (>500 pg/ml) and CRP (11.6 mg/dl) suggesting failure of amphotericin therapy. CT of the chest (Fig. 5) showed progression in bilateral bronchocentric nodular opacities and consolidations throughout the right and left upper and lower lobes as well as a slight increase in size and a cavitary lesion in the RLL. Amphotericin B was switched to posaconazole based on in vitro susceptibility.
Figure 5. CT of the chest showing progression in bilateral broncho-centric nodular opacities and consolidations
The patient continued to develop worsening respiratory failure requiring continuous oxygen supplementation. In the context of chronic immunosuppression, persistent pancytopenia, daily platelet transfusion and a poor quality of life, the patient and her family elected to transition to comfort-focused care.
DISCUSSION
Paecilomyces, an asexual fungus, is responsible for causing infection in both immunocompromised and immunocompetent individuals. Pneumonia is one such rare presentation of Paecilomyces [3]. The two clinically important species of Paecilomyces variotiis and Paecilomyces lilacinus are susceptible to different antifungal agents. Voriconazole has fungicidal activity against P. lilacinus, whereas P. variotii is more susceptible to amphotericin B and posaconazole [4].
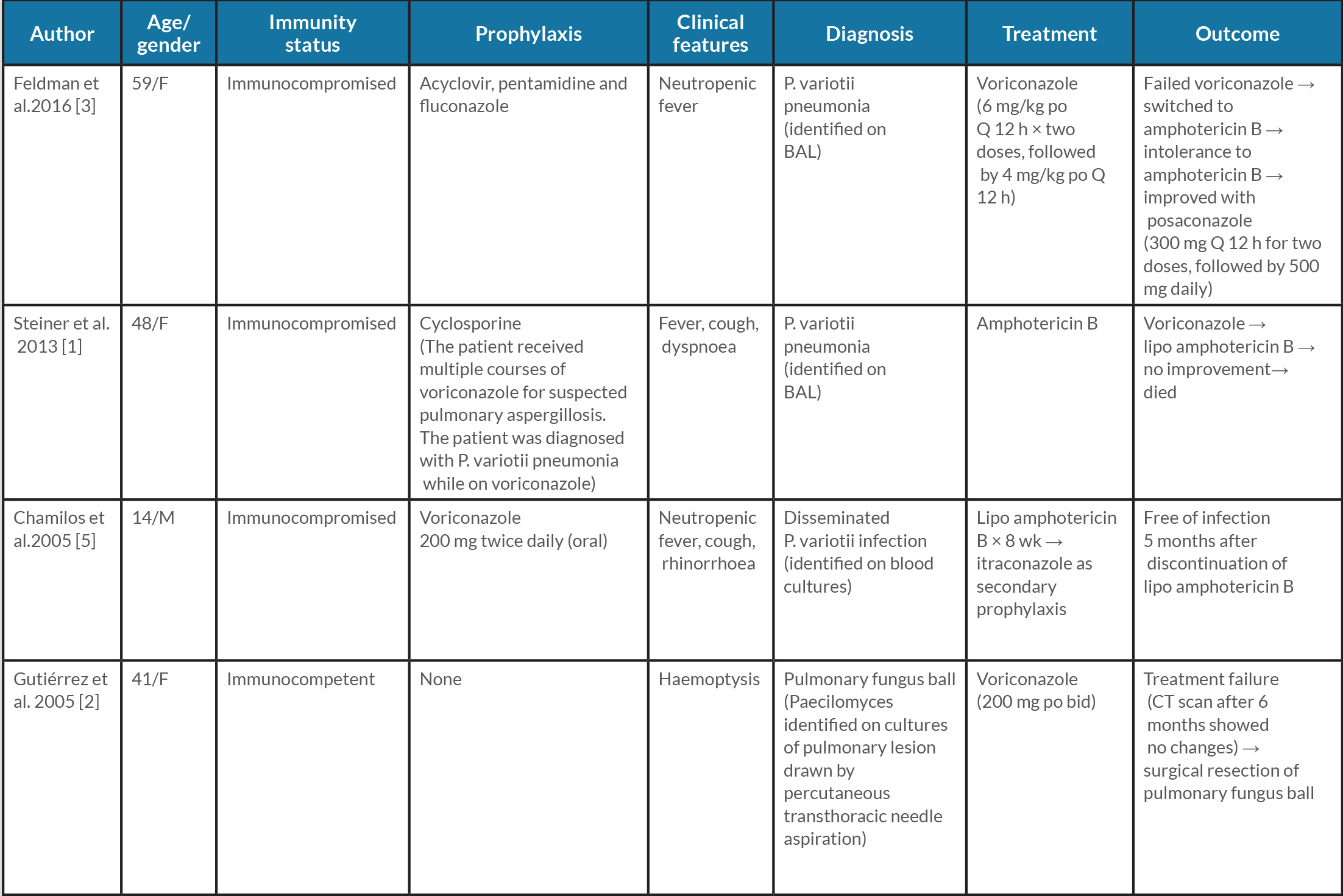
Our patient developed a rare case of Paecilomyces pneumonia despite being on voriconazole prophylaxis. In addition to our case, four other case reports have been published describing Paecilomyces infection that either failed or was resistant to voriconazole (Table 1).
Voriconazole is often the first choice of empiric therapy when prophylaxis is indicated against mould infections [3]. However, our case along with the other cases described in Table 1, bring to light the deficiency in voriconazole as empiric therapy. Posaconazole is a good alternative option for fungal prophylaxis. Its activity against both P. variotii and P. lilacinus provides an additional advantage especially in cases where speciation could not be performed [6]. Knowledge of the above literature and susceptibility testing led us to use posaconazole in our patient before the speciation could be performed.
In conclusion, Paecilomyces are a rare cause of pneumonia in immunocompromised hosts on voriconazole prophylaxis. Management of such infections is facilitated by speciation and susceptibility testing to facilitate targeted treatment.