ABSTRACT
This case highlights the importance of differentiating between Crohn’s disease and intestinal tuberculosis. The rates of misdiagnosis of Crohn’s disease and intestinal tuberculosis range from 50% to 70% because of their non-specific and clinically similar manifestations.If intestinal tuberculosis is misdiagnosed as Crohn’s disease, use of immunomodulatory drugs commonly used for Crohn’s disease can increase the risk of disseminated tuberculosis. Here we present a case highlighting the clinical similarity between these two distinct medical conditions and suggest how a similar scenario can be approached, which can help to differentiate between the two otherwise very similar conditions.
LEARNING POINTS
- Given the similarities, it is key to differentiate Crohn’s disease from intestinal tuberculosis as early as possible.
- Patients undergoing colonoscopy for possible Crohn’s disease should have colonic biopsy samples sent for AFB culture.
- Consider investigations for intestinal tuberculosis in uncontrolled Crohn’s disease where intestinal tuberculosis has not been worked up previously.
KEYWORDS
Tuberculosis, inflammatory bowel disease
CASE DESCRIPTION
A 46-year-old British-born NHS doctor of Indian descent was referred to the chest clinic for consideration of treatment for latent tuberculosis prior to the initiation of anti-tumour necrosis factor-alpha (anti TNF-α) therapy for Crohn’s disease. He had been diagnosed with Crohn’s disease 4 years earlier following the development of a perianal abscess for which he underwent surgical drainage. He had been reviewed by the gastroenterologist and was prescribed mesalazine and budesonide which he took intermittently with partial symptomatic improvement. He was considered for anti-TNF-α therapy and as a QuantiFERON® test returned positive, he was referred to the chest clinic.
In the chest clinic, the patient denied any respiratory symptoms and his chest x-ray was normal. During this consultation, symptoms, investigations and radiology were reviewed. He reported abdominal pain, cramps, loose stools and weight loss. He was born and raised in Swansea, Wales, and had no exposure to unpasteurized milk. He had no recent travel in the past 5 years. His previous perianal abscess cultures showed mixed anaerobes, so he was treated with a prolonged course of oral antibiotics. Colonoscopy revealed patchy inflammation in the transverse and ascending colon. Histological samples showed focal granuloma formations which were acid-fast (Ziehl-Neelsen) negative. A magnetic resonance enterogram reported small bowel and caecal thickening, which was thought to be consistent with Crohn’s disease.
Given the patient’s history and relatively poor response to treatment, the decision to treat for latent tuberculosis was withheld and the patient was referred for an urgent colonoscopy for an intestinal biopsy for Mycobacterium cultures, which confirmed intestinal tuberculosis, sensitive to first-line anti-tuberculosis drugs.
After discussion with the gastroenterology team, the patient’s mesalazine and budesonide were stopped and he was started on standard quadruple anti-tuberculosis treatment with complete resolution of his symptoms. A repeat magnetic resonance enterogram after intestinal tuberculosis treatment showed resolution of previous radiological changes. Due to clinical and radiological improvement, a joint decision was made not to perform any further endoscopy.
DISCUSSION
Crohn’s disease is a chronic relapsing and remitting gastrointestinal inflammatory disorder with unclear aetiology [1]. In contrast, intestinal tuberculosis can be cured with a 6-month course of chemotherapy [2]. Both diseases have many clinical, endoscopic, histological and radiological similarities [1, 2]. Correct diagnosis is important as the use of steroids, immunosuppressive agents and TNF-α therapy in Crohn’s disease may have disastrous effects on patients with tuberculosis [1, 3].
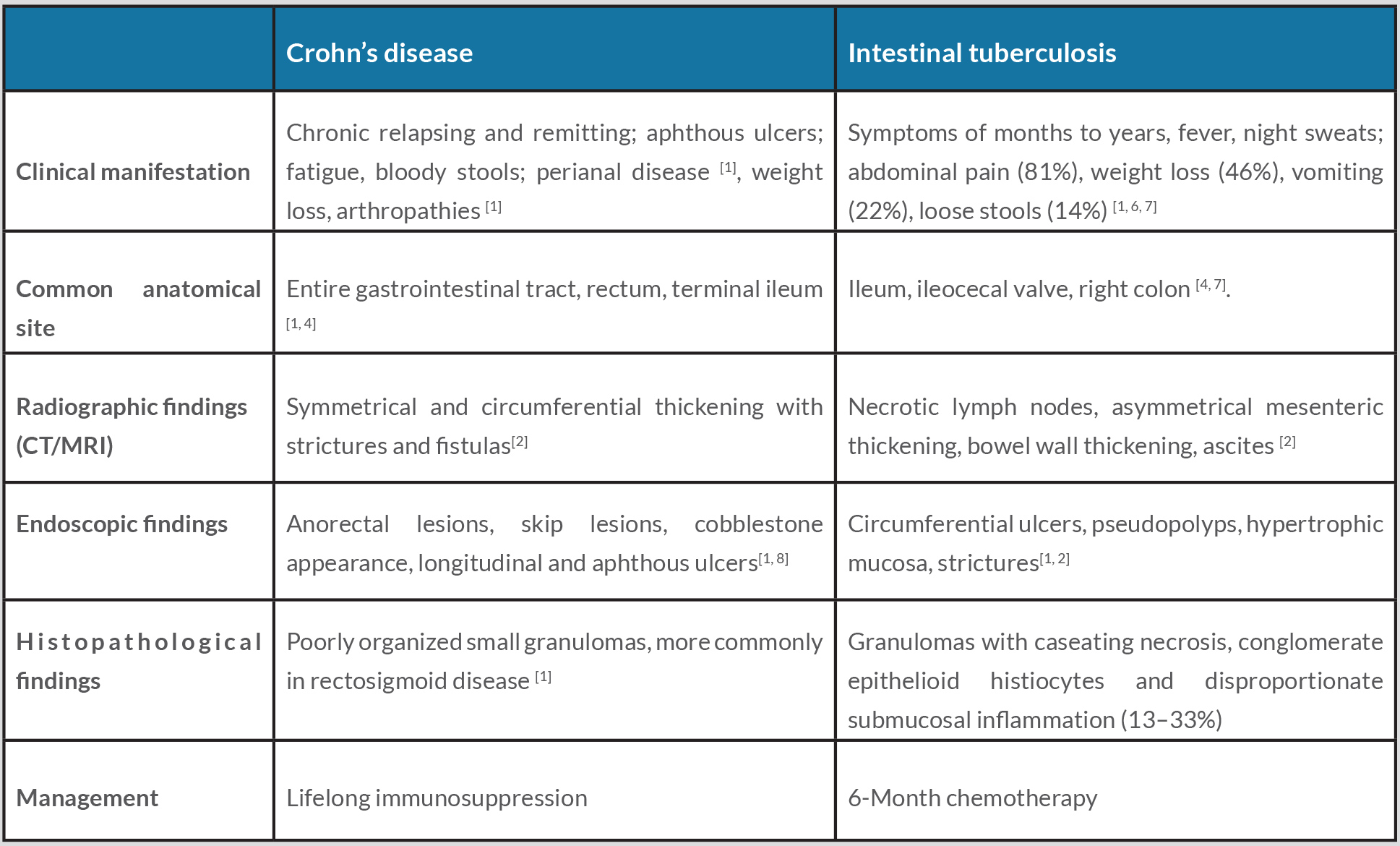
A variety of clinical, endoscopic, histological and radiological criteria have been recommended to assist with differentiating intestinal tuberculosis from Crohn’s disease (Table 1)[2, 4, 5]. Endoscopic differentiation includes anorectal lesions, a cobblestone appearance, longitudinal ulcers and aphthous ulcers, all more common in Crohn’s disease, while transverse ulcers and pseudopolyps are more common in intestinal tuberculosis [2, 5]. The endoscopic macroscopic appearance will show some overlap in 60–70% of patients [2].
Radiographically, a bowel wall with symmetrical and circumferential thickening along with strictures and fistulas is more common in Crohn’s disease, while necrotic lymph nodes are more common in intestinal tuberculosis [2, 5]. In clinical practice, however, most of these parameters lack specificity or only exist during specific stages of the diseases [4].
In intestinal tuberculosis, caseous granulomas and culture positive for Mycobacterium tuberculosis have conventionally been regarded as the gold standard for diagnosis, although often limited by poor sensitivity [1]. Granulomas with or without caseation are seen in less than 50% of resection specimens and even less frequently in biopsy samples of intestinal tuberculosis (15–36%) [1, 4].
The classic histological feature of granulomas with caseating necrosis, conglomerate epithelioid histiocytes and disproportionate submucosal inflammation are seen in only 13–33% of patients [5] and positive culture is seen in only 20–53% of cases [6]. Tuberculosis polymerase chain reaction (PCR) analysis has been shown to be more sensitive than culture and acid-fast staining in diagnosing intestinal tuberculosis, with a sensitivity of up to 66% [5] and a specificity of up to 90% [4].
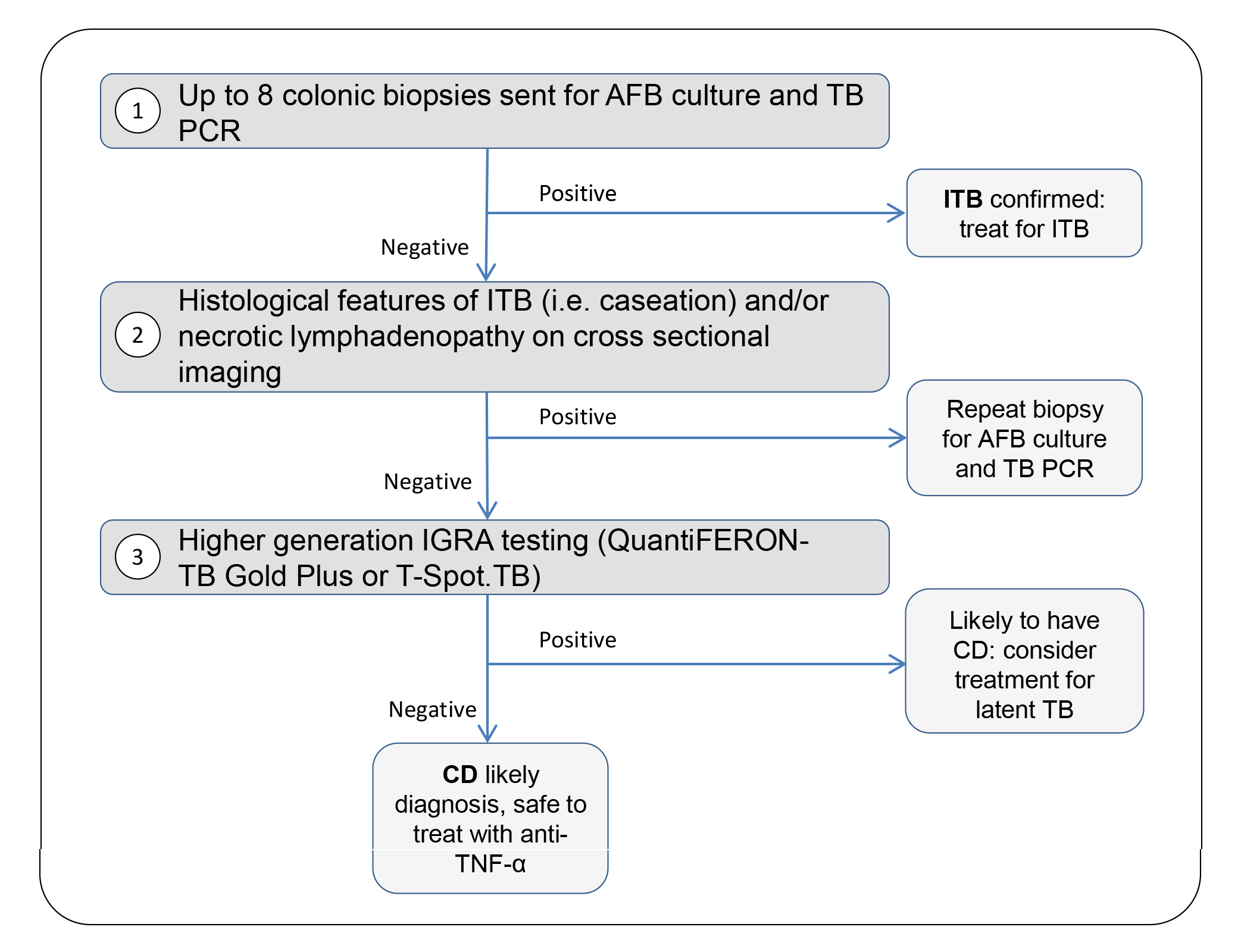
We suggest that in all patients with a clinical question of possible Crohn’s disease, the clinician perform the following steps, along with appropriate clinical assessment, to help differentiate Crohn’s disease from intestinal tuberculosis (Fig. 1): (1) send up to eight intestinal biopsy samples for acid-fast bacilli (AFB) culture [6] and if possible, tuberculosis PCR testing; (2) look for necrotic lymphadenopathy on cross-sectional imaging and histological features suggestive of intestinal tuberculosis, particularly caseation; (3) perform high generation interferon gamma release assay (IGRA) testing (QuantiFERON-TB Gold Plus or T-SPOT.TB test). A positive step 1 along with supportive clinical and/or radiological features suggestive of tuberculosis confirms intestinal tuberculosis even if the remaining two steps are not fully supportive of the diagnosis of intestinal tuberculosis. If step 1 is negative but any features of step 2 are positive, this is supportive of a diagnosis of intestinal tuberculosis. In countries with a high incidence of tuberculosis, therapeutic anti-tubercular therapy (ATT) trials might be considered at this point in patients without drug sensitivities, however we suggest repeating biopsy for cultures (step 1) to rule out drug resistance. If only step 3 is positive, the patient is more likely to have Crohn’s disease and should be considered for latent tuberculosis treatment after appropriate work-up and excluding active tuberculosis at other sites. If all three steps are negative for tuberculosis, Crohn’s disease is the likely diagnosis.
Figure 1. Proposed investigation strategy to differentiate intestinal tuberculosis (ITB) from Crohn’s disease (CD). *After appropriate work-up and excluding active TB at other sites. ITB, intestinal tuberculosis
In countries with a low incidence of tuberculosis like the United Kingdom, histological samples from patients undergoing colonoscopy for suspected Crohn’s disease are usually fixed in formalin and are not routinely sent for AFB culture. However, if latent or active tuberculosis is suspected when anti-TNF- α treatment is being considered, the patient may require another colonoscopy so histological samples can be sent for AFB culture in a non-formalin solution, as in our case. We suggest routinely sending samples in saline for AFB culture, and tuberculosis PCR if possible, for all suspected Crohn’s disease patients so the correct diagnosis can be made and a repeat procedure avoided. We believe the cost and risk of prolonging treatment to take extra specimens are minimal and offset by having the correct diagnosis and reducing the need for a repeat procedure in the future. We also suggest that patients with ‘uncontrolled’ Crohn’s disease despite treatment, where intestinal biopsies for AFB culture have not been sent previously, should be considered for repeat colonoscopy for intestinal tuberculosis work-up. We believe that such an approach may reveal some patients misdiagnosed with intestinal tuberculosis.