ABSTRACT
Deep neck space infection and viral myocarditis related to coronavirus disease 2019 (COVID-19) have both been described in the medical literature. However, there are only three reported cases of retropharyngeal infection as a presenting pathology in the setting of COVID-19.
A 26-year-old woman presented to the emergency room with fever and neck swelling and pain 1 month after COVID-19 infection. A computed tomography scan of the neck demonstrated tonsillitis with retropharyngeal infection. She was also found to have heart failure with an ejection fraction (EF) of <20% due to acute myocarditis. Her infection resolved and the EF improved to 40% prior to discharge.
Our case is the first to describe retropharyngeal infection as a late complication in an adult with a history of COVID-19 several weeks previously. It also presented a clinical challenge in terms of tailoring goal-directed medical therapy to manage severe left ventricular dysfunction caused by myocarditis.
LEARNING POINTS
- Deep neck space infections may be a long-term complication of SARS-CoV-2 infection.
- Fever, neck swelling and pain should arouse suspicion of deep neck space infection even in adults.
- Acute viral myocarditis in the setting of SARS-CoV-2 can present many weeks after the initial infection.
KEYWORDS
COVID-19, SARS-CoV-2, deep neck space infection, retropharyngeal abscess, viral myocarditis
CASE DESCRIPTION
AA 26-year-old woman presented to the hospital with a 5-day history of fever, cough and neck pain. She did not have a history of neck or head trauma, or a history of any other infection apart from SARS-CoV-2. She had tested positive for COVID-19 on a rapid antigen test 33 days previously. At that time, her symptoms were fever and myalgia, and she only received acetaminophen as needed.
On presentation to the hospital, her blood pressure was 140/94, heart rate was 134, temperature was 39.3°C, and oxygen saturation was normal. Physical examination was remarkable for pharyngeal erythema and bilateral cervical lymphadenopathy. Cardiopulmonary examination was normal.
In the emergency room, she was negative for SARS-CoV-2 on a PCR test. She had a white blood cell (WBC) count of 8.02×109/l. Her creatinine was 0.65 mg/dl. Her chest x-ray did not show any cardiopulmonary processes (Fig. 1). An electrocardiogram demonstrated sinus tachycardia with a ventricular rate of 159 (Fig. 2). Over the following 3 days, further work-up demonstrated an increase of the WBC count to 12.81×109/l, while creatinine reached 6.31 mg/dl.
Figure 1. Chest x-ray: normal cardiopulmonary anatomy.
Figure 2. ECG sinus tachycardia at 160 beats per minute.
She continued to have a heart rate above 150/minute and a temperature range of 39°C–40.5°C. Her blood pressure became labile, reaching a nadir of 84/57, which necessitated IV fluids and transfer to the intensive care unit.
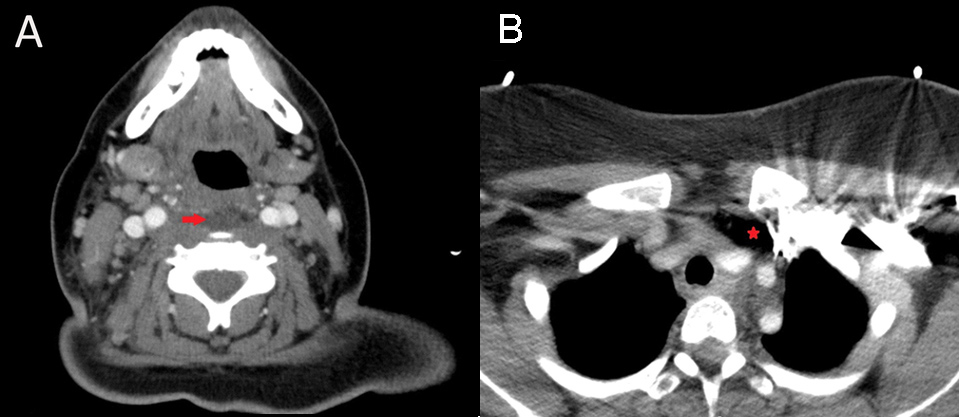
A CT scan with contrast of the neck demonstrated an enlarged right palatine tonsil, non-loculated fluid in the retropharyngeal space extending into the cervicothoracic junction, and prominent bilateral lymphadenopathy (Fig. 3). An extensive infectious investigation was carried out including blood culture, urine culture, and group A beta-haemolytic streptococcus culture, a rapid strep test, methicillin-resistant staphylococcus aureus (MRSA) swab, monospot test, parvovirus B-19 PCR, human herpesvirus-6 PCR, coxsackie B virus PCR, echovirus PCR, human immunodeficiency virus testing, and hepatitis panel, which were all negative.
The patient was evaluated by otolaryngology who recommended broad antibiotic coverage with no surgery since the fluid was non-loculated. She received piperacillin/tazobactam and IV vancomycin, which was discontinued following the negative MRSA swab. Coverage was de-escalated to ampicillin/sulbactam 7 days later. She received a total of 10 days of antibiotics.
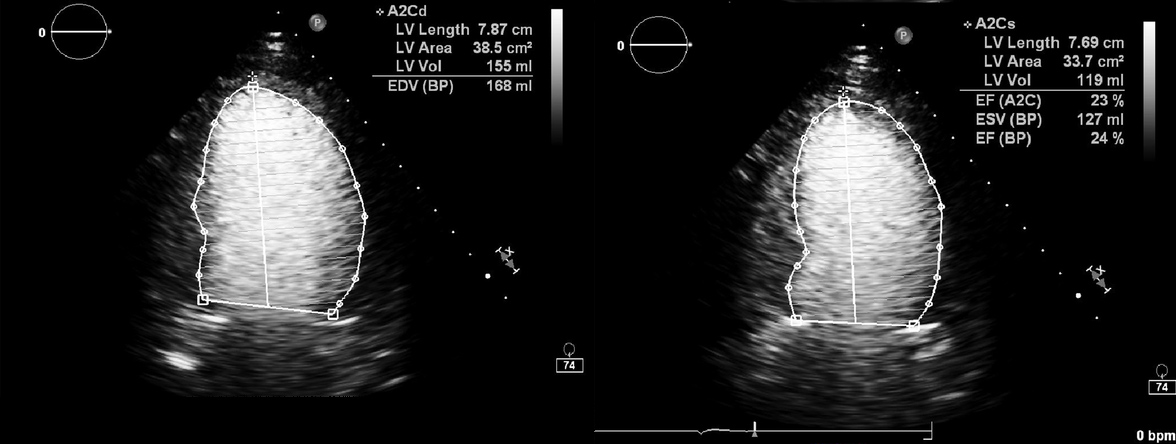
The patient had elevated troponin-I 0.87 ng/ml and brain natriuretic peptide 4925 pg/ml. An echocardiogram was carried out and showed an EF of <20%, global ventricular hypokinesia, and mild left ventricular dilation (Fig. 4). An endomyocardial biopsy and cardiac magnetic resonance imaging were deferred due to vital instability.
During her stay in the intensive care unit, the patient received increasing doses of carvedilol, rising from 3.125 mg twice a day to 25 mg twice a day while haemodynamic status was closely monitored. A second echocardiogram 9 days later showed an EF of 40% (Fig. 5). Tachycardia was better controlled with a heart rate ranging from 90 to 100, and blood pressure stabilized. She had a third echocardiogram 25 days after discharge which showed an EF of 40–45%.
Due to severe worsening of kidney function, continuous renal replacement therapy was carried out for 6 days. The patient received regular haemodialysis sessions afterwards for 3 weeks.
Figure 3. CT scan of the soft tissue of the neck with contrast: non-loculated fluid collection within the retropharyngeal region (arrow in panel-A) extending to the cervicothoracic junction (asterisk in panel-B).
Figure 4. First transthoracic echocardiogram: ejection fraction <20%.
Figure 5. Second transthoracic echocardiogram: ejection fraction 40%.
DISCUSSION
To our knowledge, there are only three reported cases of retropharyngeal infection as a presenting pathology in COVID-19 [1, 2]. Our case report is the first to describe retropharyngeal infection as a late complication in an adult with a history of COVID-19 several weeks earlier.
Retropharyngeal cellulitis is the predecessor of retropharyngeal abscess. Early detection alleviates the necessity of surgical drainage and spares patients the life-threatening consequences of further spread [3]. Expert opinion suggests antibiotic coverage against streptococci and anaerobic bacteria should be given [4, 5]. Our treatment regimen of piperacillin/tazobactam then de-escalation to ampicillin/sulbactam concurs with this approach.
There are many reports of myocarditis related to COVID-19 presenting either with concomitant infection or as a late-term complication[6, 7]. The majority of reported COVID-19 myocarditis cases are diagnosed clinically. A few cases have been confirmed by histology, and only one by histology and viral genome analysis [8]. The pathogenesis of myocarditis in COVID-19 is elusive. Notably, IL-6 is increased dramatically in patients with cardiac injury [9]. Another proposed mechanism is infection of the myocardium since SARS-CoV-2 enters human cells by binding its spike protein to angiotensin-converting enzyme 2 membrane protein [10].
We encountered a dilemma in managing the patient’s heart failure: angiotensin-converting enzyme inhibitors and angiotensin receptor blockers could not be initiated due to renal failure, and the use of beta-blockers was limited by hypotension. We chose to adopt an aggressive approach with carvedilol provided the patient’s blood pressure was not affected. In addition to its neurohormonal benefits in heart failure generally, carvedilol possesses anti-inflammatory characteristics, as shown by favourable outcomes in animal models of viral myocarditis [11, 12].