ABSTRACT
Endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) is a diagnostic tool used to investigate mediastinal lesions. It has a good safety profile, but there are rare accounts of potentially deadly complications. The present article describes one such complication: pericardial empyema. A 70-year-old man underwent EBUS-TBNA for the differential diagnosis of a pulmonary mass with multiple mediastinal adenopathies. Two weeks after the procedure he developed chest pain, shortness of breath and fever, with rapid progression to hypotension, tachycardia and low peripheral saturation. He was diagnosed with purulent pericarditis with cardiac tamponade. Pericardial drainage and antibiotic therapy were employed with successful recovery from obstructive disease and septic shock.
LEARNING POINTS
- Endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) has a good safety profile and is used to investigate peritracheal and peribronchial lesions, but severe complications have occurred so prophylactic measures should be implemented.
- Risk factors for infections due to EBUS-TBNA have not been properly established, but the presence of necrotic or cystic lesions and an elevated number of punctures during TBNA seem to be relevant.
- Although severe complications are rare, awareness of their existence may allow faster diagnosis and, consequently, a better prognosis.
KEYWORDS
Endobronchial ultrasound-guided transbronchial needle aspiration, infectious complications, pericarditis
INTRODUCTION
Endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) is a minimally invasive diagnostic method that allows the biopsy of mediastinal lymphadenopathies. In recent years it has become the standard method for investigating peritracheal and peribronchial lesions, since it has shown high sensitivity and sensitivity with a good safety profile[1]. However, its widespread use has resulted in the occurrence of rarer complications. In the literature, the incidence of complications varies from 1.4%[2] to 1.23%[3], but only 0.19%[3] are serious. One of the serious complications, pericarditis, is described in this report.
CASE DESCRIPTION
A 70-year-old male patient underwent EBUS-TBNA as part of the investigation of a pulmonary mass with multiple mediastinal adenopathies. The procedure was conducted under sedation. Two nodules were approached (#4R and #7) with four passages in each one, and the material collected showed signs of necrosis. There were no peri or postprocedural complications and the patient was discharged after 24 hours of observation. The histological diagnosis was small-cell lung cancer.
About 2 weeks later, the patient began to complain of chest pain, shortness of breath and fever. He was observed in the emergency department and admitted to the medical ward for acute kidney failure and suspicion of a respiratory infection. Despite antibiotics he developed haemodynamic instability and respiratory failure in the first 24 hours of hospitalization. Laboratory analysis showed elevated inflammation parameters (white blood cell count 24×109/l, C-reactive protein 36.45 mg/dl, procalcitonin 15.73 µ/l), hepatic cytolysis (aspartate aminotransferase 9651 U/l, alanine aminotransferase 5185 U/l), altered coagulation (prothrombin time 25 sec; INR 2.0) and worsening of acute kidney injury (creatinine 5.09 mg/dl, urea 160 mg/dl). Blood gas analysis showed respiratory failure (PaO2 40 mmHg), metabolic acidosis (pH 7.04, HCO3 9.2 mEq/l) and hyperlactacidaemia (lactate 10 mmol/l). The ECG suggested pericarditis and an echocardiogram showed pericardial effusion with cardiac tamponade physiology. The patient was admitted to the intensive care unit and subjected to emergent pericardiocentesis. Vacuum drainage was maintained for 2 days. The pericardial fluid was thick and purulent, with cytological and biochemical analysis results compatible with empyema. No malignant cells were detected. A total of 1140 ml of pericardial fluid was drained. The pericardial fluid and blood cultures were negative. Computed tomography of the chest excluded other intrathoracic infections. The patient was started on empiric antibiotic therapy with ceftriaxone and vancomycin. He had a good clinical response, recovering from all organ failures, except kidney failure. He was discharged 22 days after admission, still requiring dialysis. One month later, he died from neoplastic complications.
DISCUSSION
EBUS-TBNA is now the standard of care for the investigation of mediastinal adenopathies, and so has an important role in the diagnosis and staging of pulmonary neoplasms. It is a safe procedure, although with its increased use, complications have become more common. Pericarditis a very rare complications, with a reported incidence of 0.01%[3].
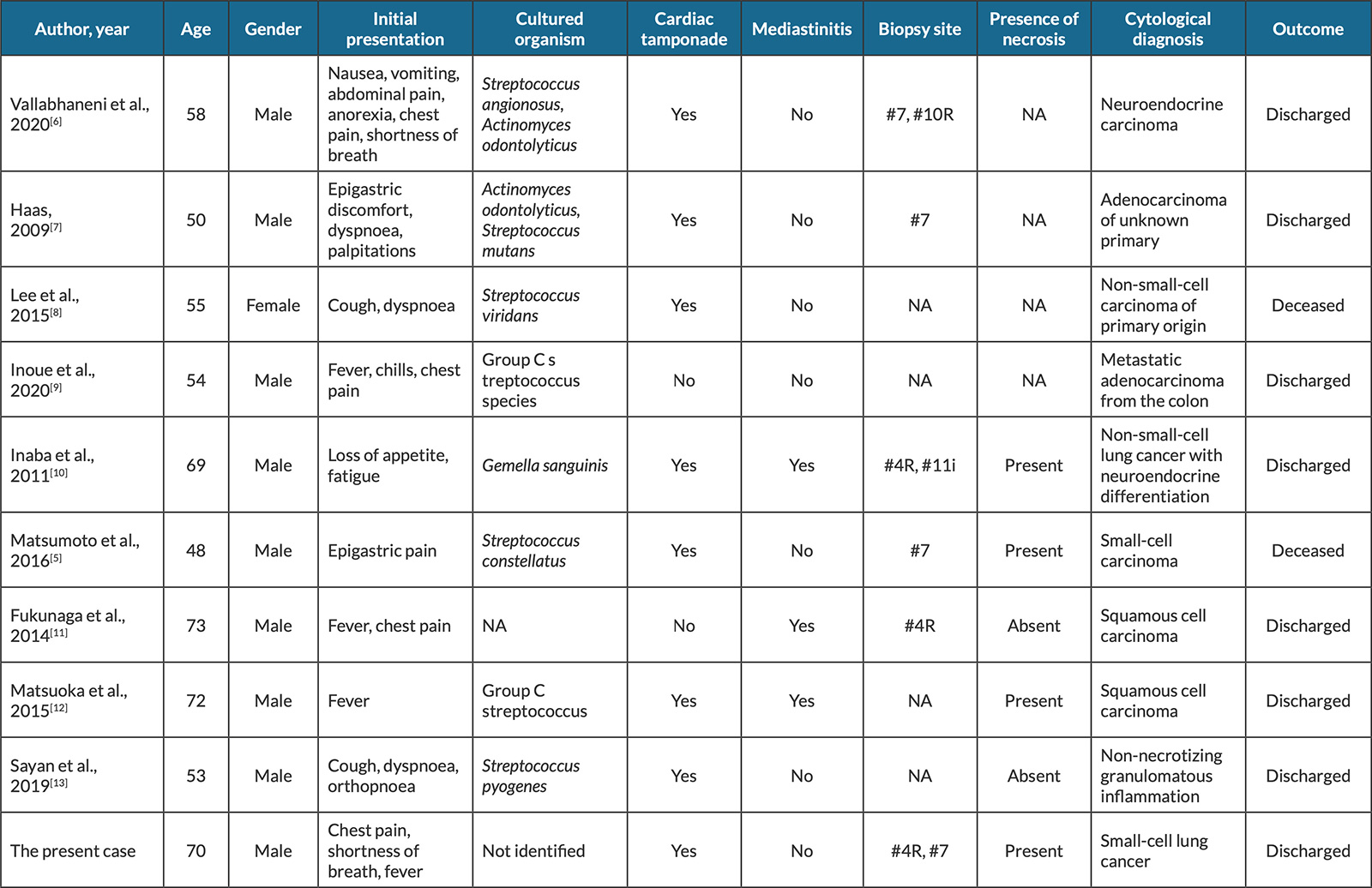
We present some information from case reports of pericarditis in the context of EBUS-TBNA in Table 1. The data show the first symptoms were unspecific and the isolated pathogens varied but came from oropharyngeal flora. Most patients with pericarditis developed cardiac tamponade, but concomitant mediastinitis was found in only a few cases. All pericardial effusions requiring drainage were purulent and most (82%) patients survived.
Table 1. Case reports of pericarditis in context of endobronchial ultrasound-guided transbronchial needle aspiration
It is thought that infectious complications are a consequence of needle contamination as the equipment passes through the oropharynx. This idea is supported by the fact that most microbiological agents isolated belong to the oropharyngeal commensal flora, and further endorsed by the clinical trial conducted by Epstein et al.[4]. In that study, all samples collected by EBUS-TBNA were subjected to microbiological culture and developed polymicrobial growth as a result.
Three pathways were proposed in the pathophysiology of pericarditis: spread of contiguous intrathoracic infection, haematogenous spread, and accidental violation of the pericardial space by the needle[5]. In our case, the pericarditis seems to have been caused by a breach of the pericardium. This hypothesis is supported by the absence of documentation of another intrathoracic infection and the proximity of the biopsy site (#4R) to the superior pericardial recess. Although the cytology and biochemical analysis of the pericardial fluid collected were compatible with empyema, the culture was negative, most likely due to the antibiotic course administered before pericardiocentesis. This fact renders the connection between the procedure and the infection more difficult, but not impossible, since a similar case was documented previously[6].
No clinical trials are described in the literature that identify the risk factors for the development of infection related to EBUS-TBNA. In some case reports, the authors suggest the biopsy of necrotic or cystic lesions may be a risk factor[3], supported by clinical knowledge of the infectious risk inherent in the biopsy of necrotic lesions in other locations. The site of the addressed lesion is also suggested as a possible risk factor, the #4R and #7 locations being the most often involved[3]. Finally, the high number of punctures during TBNA is also mentioned as a probable cause of inoculation[3].
CONCLUSIONS
EBUS-TBNA is an effective technique, with a better safety profile than its predecessor, mediastinoscopy[3]. Nevertheless, severe complications are associated with the procedure, which should be addressed so that prophylactic measures can be implemented. Patients who undergo EBUS-TBNA should be monitored carefully for signs of complications immediately afterwards and during the following weeks, since infectious complications tend to be deferred.
Risk factors for infections due to EBUS-TBNA have not been well established but the presence of necrotic or cystic lesions and an elevated number of punctures during TBNA appear to be relevant.