ABSTRACT
Sweet's syndrome or acute febrile neutrophilic dermatosis is characterized by an acute inflammatory skin eruption of oedematous and erythematous papules, plaques or nodules, accompanied by fever, and leucocytosis with possible extracutaneous involvement. Aetiologies include infections, inflammatory bowel disease, pregnancy or malignancy, or the syndrome may be drug-induced by many classes of medications or very rarely, radiocontrast exposure. Herein, the authors report a case of radiocontrast-induced bullous Sweet's syndrome and contrast-induced acute kidney injury in a woman in her 60s with a complex medical history.
LEARNING POINTS
- Patients with Sweet's syndrome (SS) typically present with acute-onset fever, leucocytosis, and erythematous, tender plaques with dense neutrophilic infiltration in the dermis. The condition is classified into three subtypes: classic SS, malignancy-associated SS, and drug-induced SS.
- Drug-induced SS is characterized by an abrupt onset of a painful erythematous rash, dense neutrophilic dermal infiltrate without vasculitis, a temporal relationship between exposure and onset, and resolution of symptoms after drug discontinuation and/or corticosteroid therapy.
- Treatment options include systemic corticosteroids as first-line therapy, while colchicine, dapsone, indomethacin, naproxen, clofazimine, ciclosporin, α-interferon, and potassium iodide may be considered as second-line therapies in cases resistant to corticosteroids.
KEYWORDS
Sweet's syndrome, acute febrile neutrophilic dermatosis, radiocontrast-induced, drug-induced, acute kidney injury, oesophageal ulcers
INTRODUCTION
Sweet's syndrome (SS) or acute febrile neutrophilic dermatosis is characterized by fever, leucocytosis with neutrophilia, and skin lesions including painful erythematous papules, nodules, and plaques caused by infiltration of neutrophils in the dermis. It can be classified into classic or idiopathic, malignancy-associated, and drug-induced SS[1]. Herein, the authors report a case of radiocontrast-induced bullous SS and contrast-induced acute kidney injury in a woman in her 60s with a complex medical history.
CASE DESCRIPTION
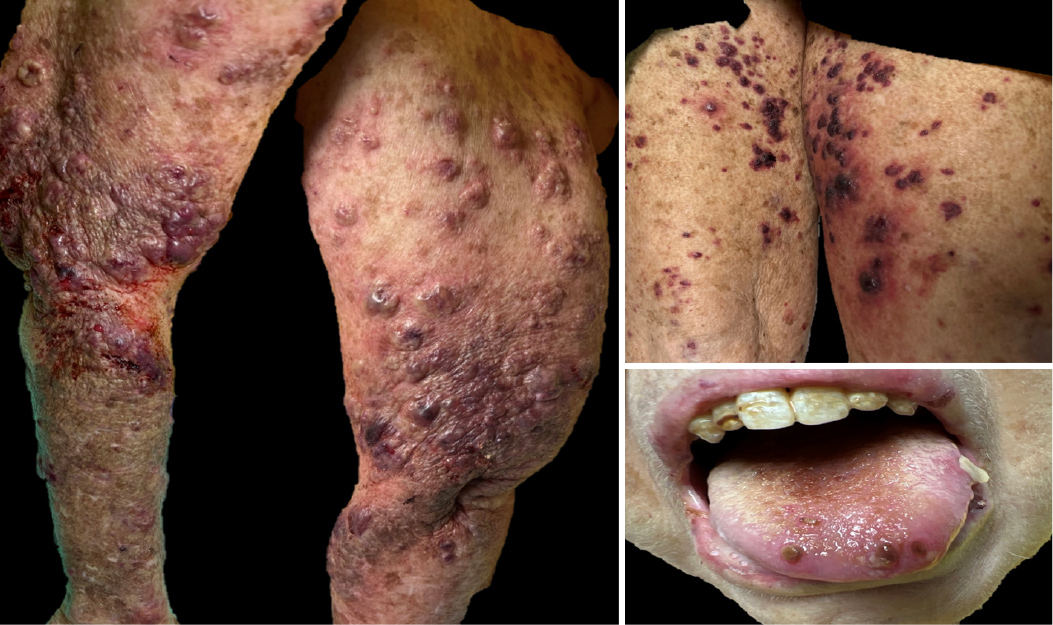
A 67-year-old woman with a medical history of hypertension, hyperthyroidism, myelodysplastic syndrome, and pyoderma gangrenosum presented for evaluation of acute onset of skin rash, 24 hours after she had undergone computed tomography (CT) scans for further evaluation of conditions associated with pyoderma gangrenosum. Upon evaluation, she complained of a progressive, painful, bloody skin rash that had started in her mouth and face and spread to her chest, arms, and legs, and which did not respond to topical emollients or topical steroids. She also complained of worsening heartburn and decreased urine output over the past 24 hours. On clinical examination, her vital signs were unremarkable with a temperature of 36.8°C, a pulse of 108/minute, blood pressure of 101/70 mmHg, respiratory rate of 16 per minute, and oxygen saturation of 98% on room air. Skin examination was significant for numerous, juicy papules with potential vesiculation, and tender haemorrhagic bullae affecting over 50% of the bilateral upper extremities with scattered foci affecting the trunk, lower extremities (inner thighs), mouth, and face (Fig. 1). Laboratory work-up was unremarkable except for normocytic normochromic anaemia with a haemoglobin of 9 g/dl (normal 12.1–15.1) and acute kidney injury with a blood urea nitrogen level of 94 mg/dl (normal 6–21), creatine level of 4.9 mg/dl (normal 0.5–1.2), and potassium level of 6.2 mmol/l (normal 3.5–5.1). Based on the history, clinical course, and physical findings, bullous SS versus the less likely toxic epidermal necrolysis was the main differential diagnosis.
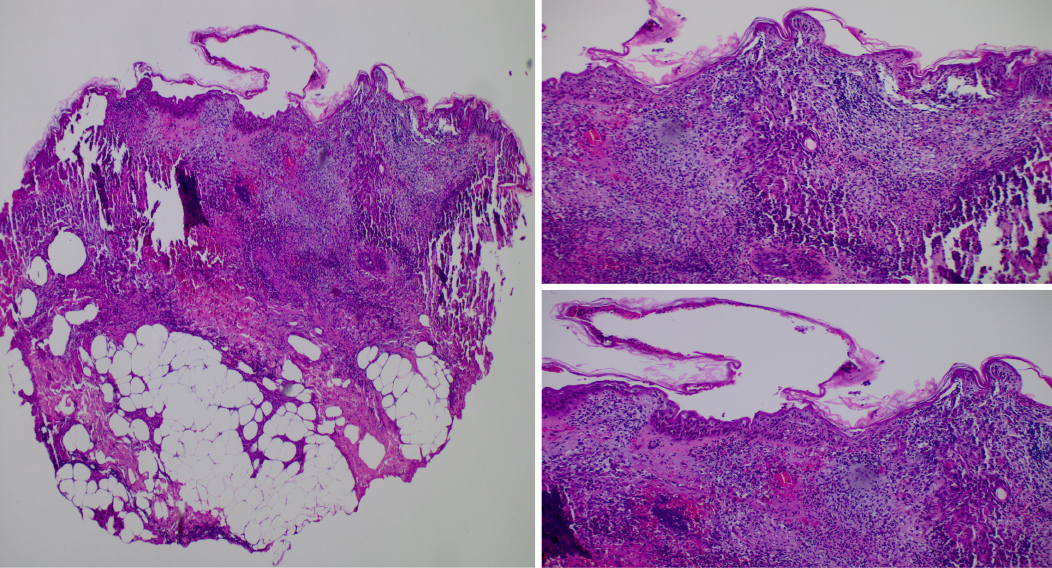
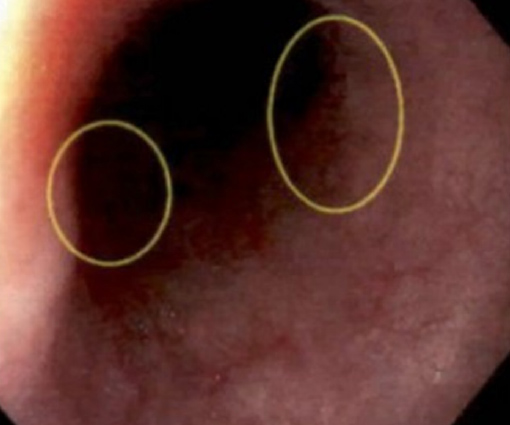
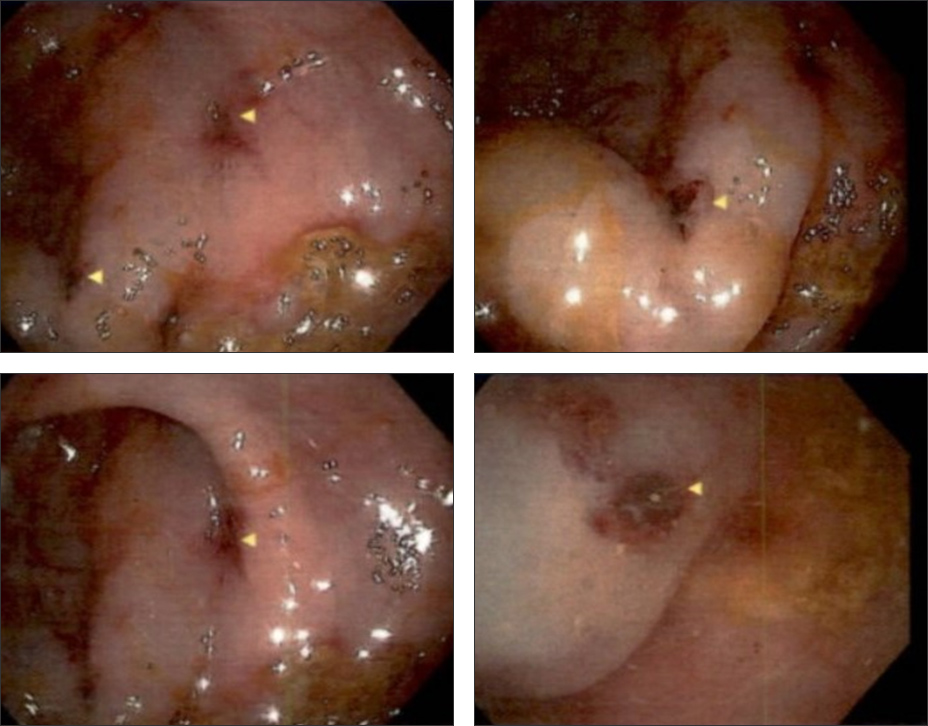
The patient underwent two 4 mm punch skin biopsies from her right upper arm. The skin biopsy samples showed diffuse neutrophilic dermatosis in the dermis. No vasculitis was noted, and the infiltration was not cantered on follicles. No organisms were seen with routine staining. Direct immunofluorescence of IgG, IgA, IgM, and C-3 tested uniformly negative, consistent with SS (Fig. 2). A diagnosis of radiocontrast-induced bullous SS was made and the patient was started on methylprednisolone 80 mg IV daily and 0.05% clobetasol ointment twice daily. Her hospital course was complicated with worsening kidney injury due to contrast-induced acute kidney injury requiring renal replacement therapy. Also, she had an episode of haematochezia for which esophagogastroduodenoscopy (EGD) and colonoscopy were performed, revealing a few superficial oesophageal ulcers with the largest measuring 4 mm in the proximal esophagus (Fig. 3) and a few 3 mm ulcers in the rectum with no active source of bleeding (Fig. 4). Biopsy results showed superficial mild and chronic inflammation with vascular congestion and focal erosion without dysplasia. The patient's condition eventually resolved, with a gradual improvement in her skin lesions after 48 hours of corticosteroid therapy. She was discharged with a tapering dose of oral prednisone therapy.
Figure 2. Skin biopsy showing diffuse neutrophilic infiltration in the dermis with no evidence of leukocytoclastic vasculitis, consistent with Sweet’s syndrome
Figure 3. Esophagogastroduodenoscopy showing oesophageal ulcers (marked by circles)
DISCUSSION
Skin manifestations related to sensitivity to radiocontrast agents are relatively common and include acute or delayed urticaria, morbilliform rash and Stevens-Johnson syndrome, while SS, originally described as ‘acute neutrophilic dermatosis', has rarely been reported as a complication following radiocontrast exposure. Neutrophilic dermatoses are a group of disorders characterized by skin lesions, histological examination of which reveals intense epidermal, dermal or hypodermal infiltrates composed primarily of neutrophils with no evidence of infection, or true vasculitis including SS, pyoderma gangrenosum, neutrophilic eccrine hidradenitis and Behçet's disease[2]. Each disease is distinguished by disease chronicity, tissue involvement, and clinical appearance, but there is some overlap in disease pathophysiology.
Classic SS is responsible for most cases, although drug-induced SS is not uncommon[3]. The most frequently reported drug associations are with granulocyte-colony stimulating factor (G-CSF), azathioprine, and all-trans retinoic acid (ATRA)[4]. The diagnostic criteria for drug-induced SS include: (a) abrupt onset of painful erythematous plaques or nodules; (b) histopathological evidence of dense neutrophilic infiltrates without evidence of leukocytoclastic vasculitis; (c) pyrexia >38°C; (d) a temporal relationship between exposure and clinical presentation; and (e) temporally related resolution of lesions after drug withdrawal or treatment with systemic corticosteroids[1]. Fever is considered the most frequent clinical sign of SS and may even precede skin lesions, but it can be absent in 10–20% of cases. Gastric ulcers have been previously reported in conjunction with Sweet's syndrome, and in this setting, contrary to typical gastric ulcers, systemic steroids may improve the skin, oral mucosa, and gastric mucosa[5]. Neutrophilia can be seen in drug-induced SS but is more common in idiopathic SS.
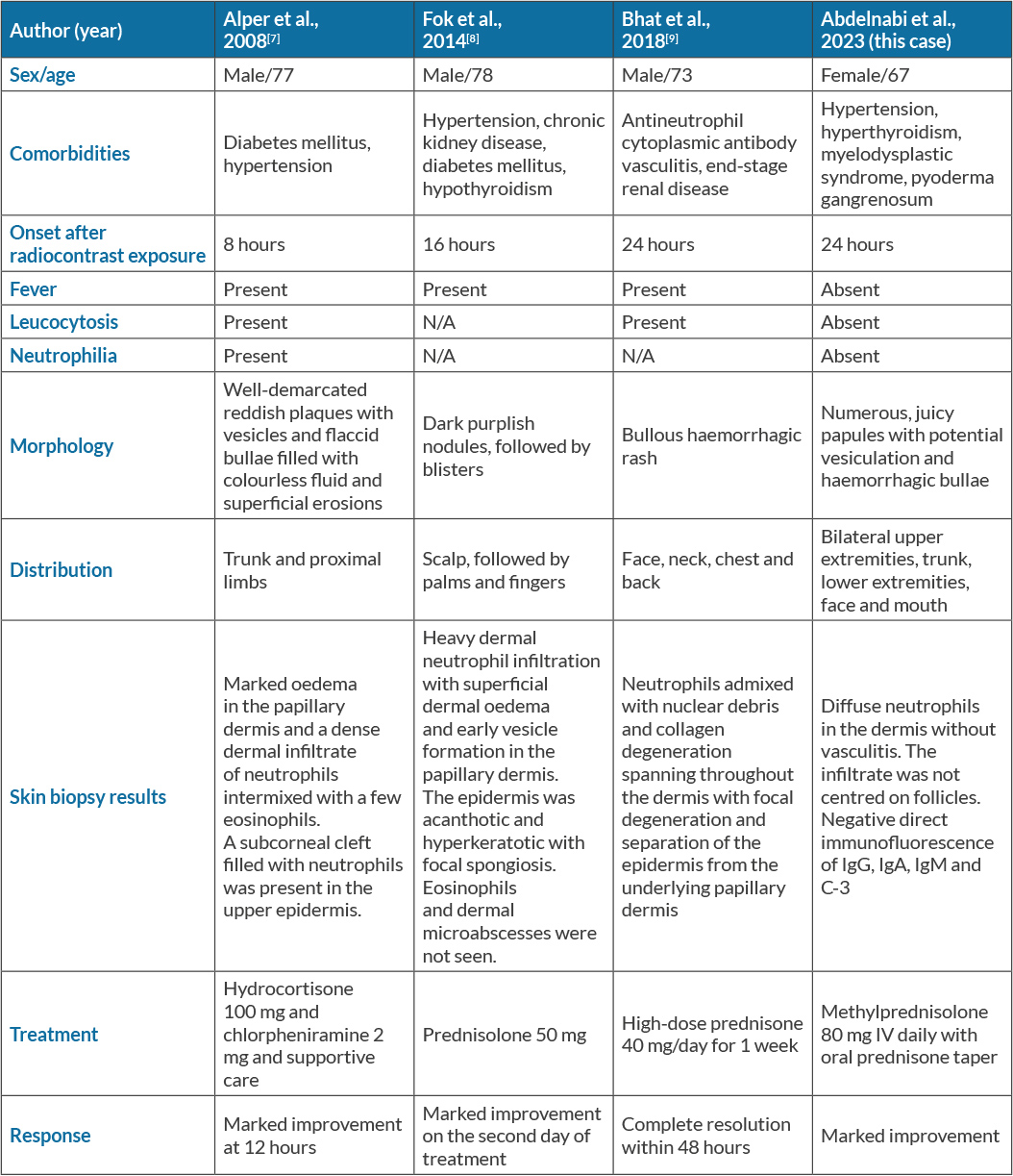
Many pathophysiological mechanisms have been proposed to explain SS. First, high levels of cytokine involvement (specifically G-CSF) promote neutrophil proliferation and maturation, and further inhibit neutrophil apoptosis as in the many cases of SS reported after the initiation of G-CSF therapy. Second, additional dysfunctional immune mediators including the adaptive immune system (specifically type 1 helper T cells with their cytokines) have been recognized as facilitating neutrophil migration and localization in the dermis. Last, exposure to stimuli causes an immune reaction to drugs and results in a cytokine cascade resulting in SS that responds to corticosteroids[3,6]. To date, only three other cases with similar presentations of radiocontrast-induced SS have been reported; these are summarized in Table 1[7–9].
Treatment for drug-induced SS consists of the administration of topical or systemic corticosteroids together with withdrawal of the drug. The average time until skin lesions improved with systemic treatment was 8 days. The time for clinical improvement with topical treatment varied from 3 to 30 days. In rare cases of corticosteroid failure, alternative therapies such as colchicine, dapsone, indomethacin, naproxen, clofazimine, ciclosporin, α-interferon, and potassium iodide may be utilized. Recurrence is uncommon in drug-induced SS after discontinuation of the medication[4].
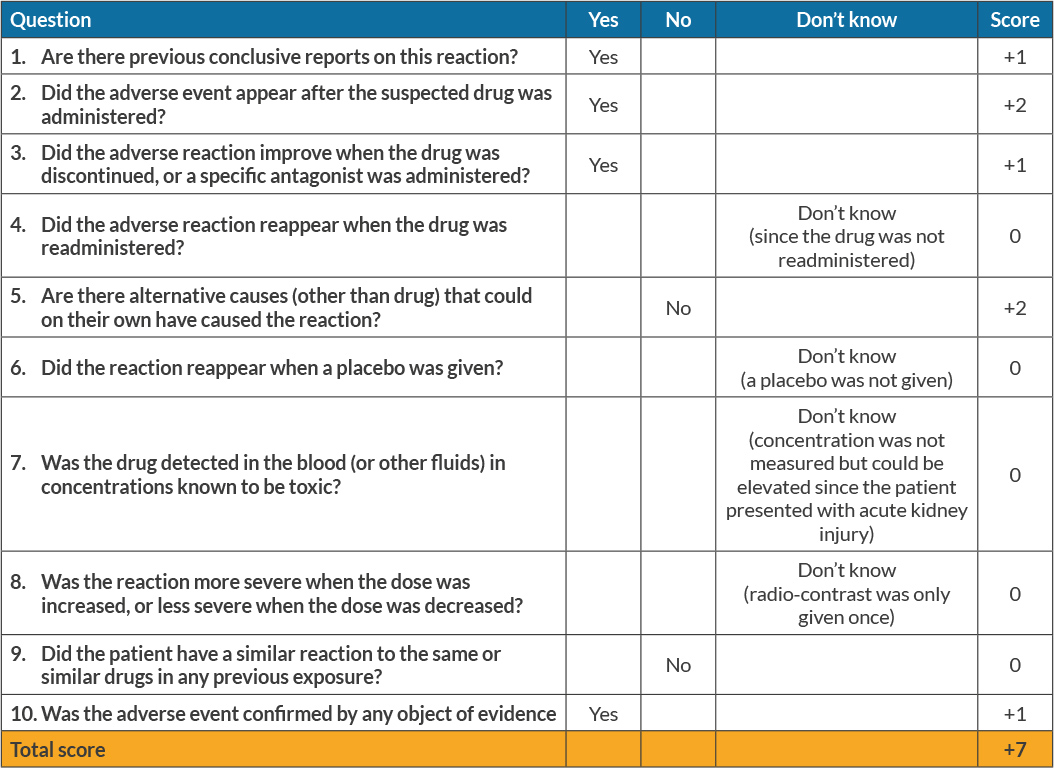
The authors have presented a case of radiocontrast-induced bullous SS and contrast-induced acute kidney injury in a woman with a complex medical history. There was a direct temporal relationship between contrast exposure and the abrupt onset of bullous skin lesions. A review of her medications showed she had not been previously exposed to any drugs associated with SS. Local bacterial infection of her skin lesions was also ruled out by negative tissue cultures. De novo or recurrent malignancies such as haematological malignancies or solid tumours such as adenocarcinoma[1] were excluded by negative CT scans. A skin biopsy confirmed the diagnosis and the skin lesions improved with corticosteroid therapy and discontinuation of the offending agent. The Adverse Drug Reactions Probability Scale[10] score was 7, indicating a probable association between exposure to radiocontrast and the onset of SS (Table 2). However, there were some limitations to this calculation as applied to our patient since some of the parameters could not be tested, such as whether the adverse reaction reappeared if the contrast was readministered or with placebo, whether the adverse reaction was more or less severe with higher or lower doses, or whether it was detected in known toxic concentrations.
Consequently, the score might have been falsely low; nevertheless, our patient met all five diagnostic criteria for drug/contrast-induced SS[11].
CONCLUSIONS
This report has described a case of radiocontrast-induced haemorrhagic bullous SS associated with contrast-induced acute kidney injury. History, clinical examination with abrupt onset of a painful erythematous rash, dense neutrophilic dermal infiltrate without vasculitis after drug/contrast exposure, and resolution of symptoms after urgent cessation and early initiation of high-dose steroids all suggested a diagnosis of SS.